Scientists Discover Better Way To Capture Carbon From Industrial Emissions
[ad_1]

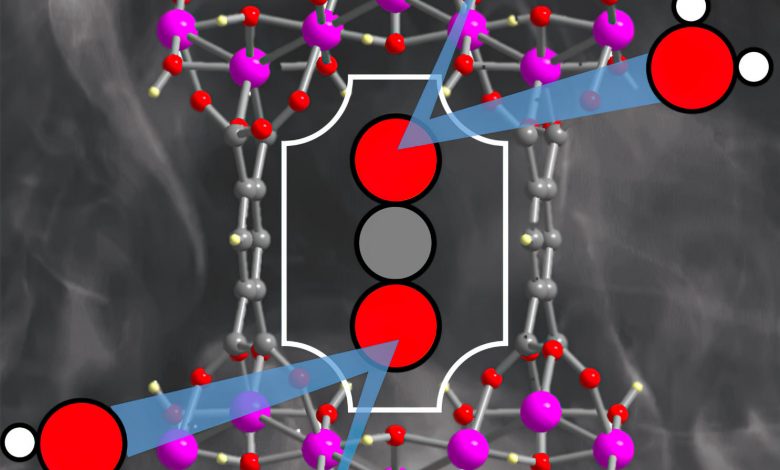
Researchers from Oregon State University have discovered the potential of a cost-effective nanomaterial, known as a metal-organic framework (MOF), to efficiently remove carbon dioxide from industrial emissions, even in damp conditions. The new MOF, composed of aluminum and a common ligand, offers a promising solution to some of the challenges in carbon capture, including high costs and reduced effectiveness in humid environments. Credit: Image provided by Kyriakos Stylianou, OSU College of Science.
Scientists from the College of Science at Oregon State University have showcased the potential of a cost-effective nanomaterial to filter carbon dioxide from industrial pollutants.
The findings, published in Cell Reports Physical Science, are important because improved carbon capture methods are key to addressing climate change, said OSU’s Kyriakos Stylianou, who led the study.
Carbon dioxide, a greenhouse gas, results from burning fossil fuels and is one of the primary causes of a warming climate.
Facilities that filter carbon from the air are beginning to spring up around the globe – the world’s largest opened in 2021 in Iceland – but they’re not ready to make a large dent in the worldwide emissions problem, Stylianou notes. In a year, the Iceland plant can draw out a carbon dioxide amount equivalent to the annual emissions of about 800 cars.
However, technologies for mitigating carbon dioxide at the point of entry into the atmosphere, such as a factory, are comparatively well developed. One of those technologies involves nanomaterials known as metal-organic frameworks, or MOFs, that can intercept carbon dioxide molecules through adsorption as flue gases make their way through smokestacks.
“The capture of carbon dioxide is critical for meeting net-zero emission targets,” said Stylianou, an assistant professor of chemistry. “MOFs have shown a lot of promise for carbon capture because of their porosity and their structural versatility, but synthesizing them often means using reagents that are costly both economically and environmentally, such as heavy metal salts and toxic solvents.”
In addition, dealing with the water portion of smokestack gases greatly complicates removing the carbon dioxide, he said. Many MOFs that have shown carbon capture potential lost their effectiveness in humid conditions. Flue gases can be dried, Stylianou said, but that adds significant expense to the carbon dioxide removal process, enough to make it nonviable for industrial applications.
“So we sought to come up with a MOF to address the various limitations of the materials currently used in carbon capture: high cost, poor selectivity for carbon dioxide, low stability in humid conditions, and low CO2 uptake capacities,” he said.
MOFs are crystalline, porous materials made up of positively charged metal ions surrounded by organic “linker” molecules known as ligands. The metal ions make nodes that bind the linkers’ arms to form a repeating structure that looks something like a cage; the structure has nanosized pores that adsorb gases, similar to a sponge.
MOFs can be designed with a variety of components, which determine the MOF’s properties, and there are millions of possible MOFs, Stylianou said. Almost 100,000 of them have been synthesized by chemistry researchers, and the properties of another half-million have been predicted.
“In this study, we introduce a MOF composed of aluminum and a readily available ligand, benzene-1,2,4,5-tetracarboxylic acid,” Stylianou said. “The synthesis of the MOF happens in water and only takes a couple of hours. And the MOF has pores with a size comparable to that of CO2 molecules, meaning there’s a confined space for incarcerating the carbon dioxide.”
The MOF works well in damp conditions and also prefers carbon dioxide to nitrogen, which is important because nitrogen oxides are an ingredient in flue gasses. Without that selectivity, the MOF would potentially be binding to the wrong molecules.
“This MOF is an outstanding candidate for wet post-combustion carbon capture applications,” Stylianou said. “It’s cost-effective with exceptional separation performance and can be regenerated and reused at least three times with comparable uptake capacities.”
Reference: “CO2 capture from wet flue gas using a water-stable and cost-effective metal-organic framework” by Ryan P. Loughran, Tara Hurley, Andrzej Gładysiak, Arunraj Chidambaram, Konstantin Khivantsev, Eric D. Walter, Trent R. Graham, Patrick Reardon, Janos Szanyi, Dylan B. Fast, Quin R.S. Miller, Ah-Hyung Alissa Park, and Kyriakos C. Stylianou, 30 June 2023, Cell Reports Physical Science.
DOI: 10.1016/j.xcrp.2023.101470
Scientists from Columbia University, the Pacific Northwest National Laboratory and Chemspeed Technologies AG of Switzerland also took part in this research, as did Oregon State chemists Ryan Loughran, Tara Hurley and Andrzej Gładysiak.
The Oregon State College of Science, the OSU Department of Chemistry, and Brian and Marilyn Kleiner supported the study.
[ad_2]
Source link